Odoo ERP for Medical Device Manufacturers: Equipment & Inventory Management
- April 21, 2026
- Posted by: Abinay Subramaniam P
- Categories: Blog, Odoo ERP for medical device manufacturers
Why the Medical Device and Hospital Supply Chain Is Uniquely Complex
Compliance gaps tied to frameworks such as FDA 21 CFR Part 820, ISO 13485, and EU MDR expose organizations to regulatory action, legal exposure, and substantial revenue loss. Asset downtime inside hospitals compounds the impact of delayed procedures, rescheduled care, and reputational damage quickly.
This is not a linear supply chain. It is a tightly coupled system where manufacturing precision, regulatory discipline, and hospital operations intersect. That is why organizations are shifting toward medical device manufacturing software supported by medical device erp software and ERP for medical device manufacturers to maintain traceability, enforce quality controls, and sustain operational continuity across stakeholders.
The Invisible Thread: From Factory Floor to Patient Bedside
A deviation during production component variance, calibration drift, or documentation lapse can propagate downstream into clinical environments. Without end-to-end visibility, the root cause remains buried until a patient-safety incident surfaces. With medical device manufacturing software integrated into a medical device ERP system, manufacturers can link design history records, batch data, and quality events to distribution pathways. On the hospital side, medical equipment management software and medical equipment inventory management software connect device usage, maintenance logs, and asset performance. This continuity closes the loop between manufacturer accountability and hospital safety outcomes.
Regulatory Pressure Is Constant and Non-Negotiable
Compliance is not a checkpoint; it is an ongoing discipline. Regulations such as FDA 21 CFR Part 820, ISO 13485, EU MDR, and UDI demand traceability, audit readiness, and documented quality controls across the product lifecycle. Generic platforms fall short because they cannot enforce device history tracking, complaint handling workflows, or audit trails at the level regulators expect. In contrast, medical device erp software aligned with erp for medical device manufacturers embeds compliance into daily operations capturing data at source, maintaining validation records, and supporting inspection readiness without disruption.
Why Generic Software Fails in Regulated Healthcare
Conventional ERP tools prioritize transactional efficiency, not regulated traceability. They lack:
- Device-level genealogy and batch traceability
- Integrated quality management tied to production events
- Calibration, maintenance, and service lifecycle tracking
- Audit-ready documentation aligned with regulatory standards
Without these capabilities, organizations rely on fragmented systems and spreadsheets introducing risk, delays, and data inconsistencies. Medical device manufacturing software combined with medical device erp system architecture resolves this gap by aligning production, quality, and compliance into a single operational framework. On the provider side, medical equipment inventory management software ensures asset visibility, utilization tracking, and maintenance accountability across departments.
Who This Guide Serves:
This guide is designed for professionals responsible for outcomes across the value chain:
- Quality managers safeguarding compliance and audit readiness
- Hospital operations leaders responsible for uptime and clinical continuity
- Procurement directors managing vendor reliability and cost exposure
- IT teams tasked with system integration, validation, and data governance
Each role depends on accurate, connected data delivered through medical device manufacturing software, medical equipment management software, and medical device erp software working in alignment.
What This Guide Will Unpack
The sections ahead examine the full software landscape shaping this ecosystem:
- How medical device manufacturing software governs production quality and traceability
- The role of medical device erp system platforms in integrating finance, supply chain, and compliance
- How medical equipment management software supports hospital asset lifecycle control
- Why medical equipment inventory management software is central to utilization, maintenance, and risk reduction
- How erp for medical device manufacturers connects upstream production with downstream clinical impact
This is a connected environment. Success depends on visibility across the chain from raw material to patient outcome supported by systems designed for regulated healthcare, not adapted after the fact.
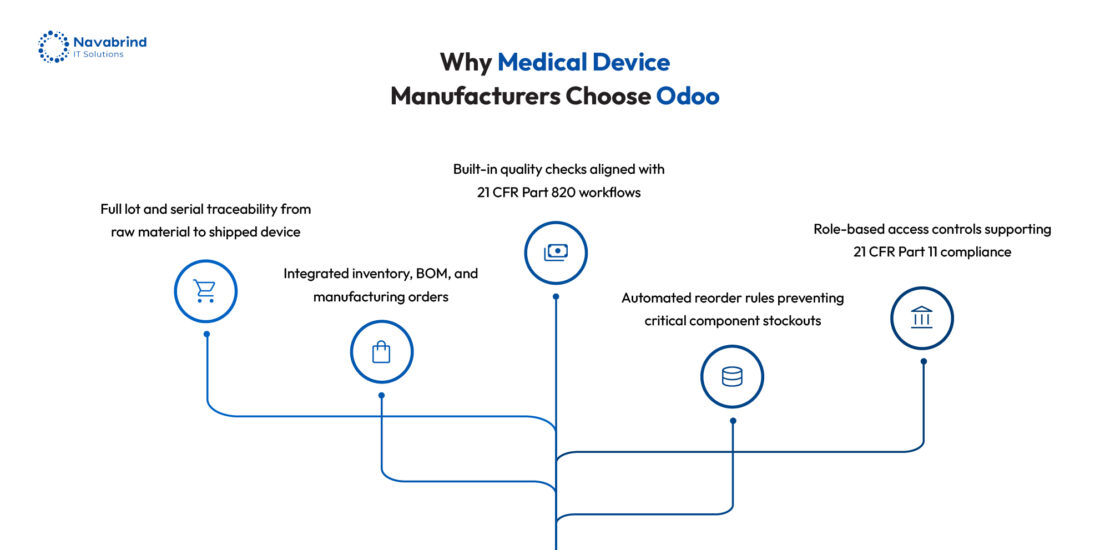
Medical Device Manufacturing Software — What It Is and What It Must Do
Medical device manufacturing software defines a tightly governed production environment engineered for regulated device fabrication. Unlike general manufacturing tools, this category operates as a Manufacturing Execution System (MES) aligned with compliance mandates, traceability requirements, and quality enforcement across the production lifecycle. Standard manufacturing platforms focus on throughput and cost control. In contrast, medical device manufacturing software working alongside medical device erp software and erp for medical device manufacturers anchors production in documented quality, validated processes, and audit-ready data integrity.
What Sets Medical Device Manufacturing Software Apart
At its foundation, medical device manufacturing software manages execution on the shop floor with full traceability and compliance alignment. Each unit, batch, and component is tracked across production stages, ensuring accountability from raw material intake to finished device release. This level of control integrates seamlessly with a medical device erp system, allowing organizations to synchronize production, procurement, and compliance records without data fragmentation.
Must-Have Capabilities That Define the Category
1. Lot and Batch Traceability:
Traceability is non-negotiable. The system must map materials, components, and finished devices across the lifecycle. In the event of a defect, teams can isolate impacted batches without disrupting unaffected inventory. This capability strengthens recall management and supports regulatory expectations.
2. Non-Conformance and Deviation Management:
Production deviations must trigger controlled workflows capturing root cause, corrective action, and preventive measures. Medical device manufacturing software ensures that non-conformance events are logged, reviewed, and resolved within compliance boundaries.
3. DHR and DHF Generation:
Device History Records (DHR) and Device Master Files (DHF) are generated and maintained within the system. This documentation forms the backbone of audit readiness, linking production data with design and quality records.
4. Electronic Batch Records and E-Signatures:
Digital batch records replace paper-based logs, supported by compliant e-signatures aligned with 21 CFR Part 11. This ensures data integrity, user accountability, and secure recordkeeping across manufacturing operations.
5. Integration with Quality and Laboratory Systems:
Production cannot operate in isolation. Medical device manufacturing software integrates with QMS platforms and laboratory data systems, connecting inspection results, validation data, and quality checks with manufacturing workflows. This integration is critical for maintaining compliance and ensuring consistent product quality.
Compliance Is Embedded Into the Workflow
Good Manufacturing Practice (GMP) is enforced at the system level. Instead of adding compliance layers later, medical device manufacturing software embeds process controls, validation checkpoints, and audit trails into daily operations. This ensures that each production step aligns with regulatory frameworks such as FDA 21 CFR Part 820 and ISO 13485. Through integration with a medical device erp system, organizations maintain a continuous chain of compliant data from procurement through production to distribution.
Deployment Models and Validation Considerations
Deployment strategy influences validation requirements:
- On-premise environments provide tighter infrastructure control, often preferred for organizations with internal validation teams and strict data governance policies.
- Cloud-based environments offer scalability and faster rollout cycles, though validation must align with Computer System Validation (CSV) and Computer Software Assurance (CSA) expectations.
Regardless of deployment model, medical device ERP software and medical device manufacturing software must maintain validated states, controlled updates, and documented change management to satisfy regulatory scrutiny.
Case Snapshot: Measurable Impact After Implementation
A mid-sized device manufacturer transitioned from fragmented systems to an integrated medical device manufacturing software platform connected with a medical device erp system.
Within months:
- Batch traceability improved, enabling rapid isolation of quality issues
- Audit preparation timelines reduced through structured DHR and DHF documentation
- Deviation handling cycles shortened with enforced workflows
- Production visibility increased across departments, strengthening coordination with hospital procurement teams relying on medical equipment inventory management software
The result is stronger compliance posture, reduced operational risk, and measurable gains in production reliability. Medical device manufacturing software is not an optional upgrade. It forms the operational backbone for organizations that must deliver safe, compliant, and traceable medical devices at scale supported by medical device erp software, erp for medical device manufacturers, and connected hospital systems such as medical equipment management software.
ERP for Medical Device Manufacturers: The System of Record That Ties It All Together
Medical device erp software functions as the operational backbone for regulated manufacturers. Beyond accounting, this system governs product data, quality workflows, supplier performance, and compliance documentation across the lifecycle. When aligned with medical device manufacturing software, the medical device ERP system becomes the single source of governed data connecting production, quality, and financial control in one validated environment. Organizations relying on generic ERP platforms face structural gaps. Standard deployments of SAP, Oracle, or NetSuite lack native support for device traceability, regulatory documentation, and audit-grade workflows. Retrofitting these systems introduces risk, fragmented data, and validation challenges. In contrast, erp for medical device manufacturers embeds compliance logic into operational processes from the ground up.
Core ERP Capabilities for Medical Device Operations:
1. Bill of Materials (BOM) and Product Structure Control
The medical device ERP system manages multi-level BOMs with version control, ensuring traceability across design revisions and production batches. Each component is linked to supplier data, inspection records, and compliance documentation.
2. Supplier Quality and Performance Management
Approved vendor lists, qualification workflows, and supplier audits are maintained within medical device erp software. Incoming inspection results are captured against purchase orders, ensuring that only compliant materials enter production. Supplier corrective actions are tracked with full accountability.
3. CAPA (Corrective and Preventive Actions)
CAPA workflows are embedded within the ERP environment, linking quality events from medical device manufacturing software to investigation, resolution, and preventive measures. This ensures that deviations translate into controlled, documented improvements.
4. Change Control and Engineering Governance
Engineering changes must follow controlled approval cycles. The ERP system enforces versioning, impact analysis, and release controls, ensuring that product updates align with compliance expectations and production readiness.
UDI Compliance Across the Product Lifecycle
Unique Device Identification (UDI) requirements are enforced through UDI frameworks. The medical device erp system manages device identifiers, production identifiers, and labeling data across manufacturing and distribution.
This capability ensures:
- Accurate labeling and serialization
- Traceability across distribution channels
- Alignment with global regulatory databases
When integrated with medical device manufacturing software, UDI data flows from production to packaging and shipment without data gaps.
Financial Control Linked to Compliance
Financial visibility in medical device erp software extends beyond standard accounting. The system supports:
- Standard costing aligned with production processes
- Lot-level cost tracking for traceability and margin analysis
- Warranty provisions based on product performance data
- Recall accruals linked to quality risk exposure
This integration ensures that financial reporting reflects operational realities, including compliance-driven costs and risk exposure.
Supplier Management as a Compliance Function
Supplier relationships carry regulatory implications. The medical device erp system enforces:
- Approved vendor qualification and re-evaluation
- Incoming inspection tied to procurement workflows
- Supplier corrective action tracking
- Performance monitoring aligned with quality benchmarks
This transforms procurement into a governed process, tightly aligned with medical device manufacturing software and downstream hospital requirements supported by medical equipment inventory management software.
Industry-Specific ERP vs. Configured General ERP — A Decision Framework
Configured General ERP:
- Requires extensive customization for compliance workflows
- Higher validation overhead due to custom code
- Increased risk of inconsistencies across modules
Industry-Aligned Medical Device ERP:
- Pre-aligned with regulatory frameworks such as FDA 21 CFR Part 820 and ISO 13485
- Faster validation cycles due to standardized processes
- Integrated quality, traceability, and documentation controls
For regulated manufacturers, erp for medical device manufacturers reduces operational risk and accelerates compliance readiness without excessive system modification.
The Validated Data Ecosystem: ERP at the Center
The medical device erp system connects with surrounding systems to form a controlled data environment:
- ERP ↔ Medical Device Manufacturing Software (MES): production execution, batch data, traceability
- ERP ↔ QMS: quality events, CAPA workflows, audit records
- ERP ↔ LIMS: laboratory testing, validation data, inspection results
This integration ensures that data flows across systems with consistency and audit integrity. Each transaction whether production, quality, or financial remains traceable and verifiable. Medical device erp software is not a back-office tool. It is the governing layer that aligns production, compliance, and financial control working in tandem with medical device manufacturing software, medical equipment management software, and medical equipment inventory management software to sustain performance across the healthcare value chain.
Medical Device Compliance Software: Staying Ahead of Regulators, Not Chasing Them
Compliance in the device sector demands continuous control, documented evidence, and audit-ready systems. Medical device compliance software operates alongside medical device erp software and medical device manufacturing software, forming a dedicated layer focused on governance, risk, and regulatory alignment. This is not an extension of ERP, a specialized environment designed to manage quality systems, regulatory documentation, and post-market obligations with discipline. Manufacturers investing in standalone compliance platforms gain control over regulatory exposure, reduce audit risk, and strengthen market access across regions.
The Compliance Landscape in 2025–2026:
Regulatory expectations continue to tighten across global markets. The transition to EU MDR has raised the bar for clinical evidence, traceability, and post-market monitoring. In the United States, the FDA QMSR aligns device quality systems with international standards such as ISO 13485. For manufacturers targeting multi-region distribution, compliance software ensures alignment across regulatory frameworks without duplicating effort. This capability supports faster approvals, sustained certifications, and reduced risk of market withdrawal.
What Compliance Software Automates:
- Document Control and Version Governance:
Policies, procedures, and technical files are managed with strict version control, approval workflows, and audit tracking. This ensures that only validated documents guide production and quality processes. - Training and Competency Records:
Employee training is tracked against roles and regulatory requirements. Compliance software enforces qualification before personnel engage in regulated activities, maintaining accountability across teams. - Audit Management:
Internal and external audits are planned, executed, and documented within the system. Findings, observations, and corrective actions are recorded with full traceability. - CAPA Workflows:
Corrective and preventive actions are initiated from quality events, complaints, or audit findings. These workflows integrate with medical device erp system and medical device manufacturing software, ensuring that issues identified in production or field usage translate into controlled remediation.
Risk Management Integrated Across the Lifecycle
Risk management aligned with ISO 14971 is embedded within compliance platforms. Risk files are linked to design inputs, production data, and post-market feedback.
This integration ensures:
- Risk identification during design phases
- Continuous risk evaluation during production
- Feedback loops from field data into risk controls
When connected with erp for medical device manufacturers, risk data influences supplier selection, process controls, and product updates.
Post-Market Surveillance and Field Intelligence
Regulators expect manufacturers to monitor device performance beyond release. Compliance software manages:
- Complaint intake and investigation workflows
- Medical Device Reporting (MDR) and vigilance reporting
- Signal detection based on complaint trends and failure patterns
This capability ensures that emerging risks are identified early and addressed before escalation into large-scale recalls or regulatory intervention.
Audit Trails and Electronic Records
Inspection readiness depends on data integrity. Systems must comply with 21 CFR Part 11, ensuring:
- Secure electronic records
- Controlled user access and authentication
- Time-stamped audit trails for all actions
These controls are enforced within compliance platforms and synchronized with medical device erp software, creating a validated and traceable data environment.
ROI of Compliance Software: Prevention vs. Exposure
The financial impact of non-compliance is measurable. A single recall can result in:
- Product withdrawal costs
- Regulatory penalties
- Legal exposure
- Loss of market trust
In contrast, investment in medical device compliance software reduces:
- Audit preparation time
- Recall frequency through early detection
- Cost associated with rework and non-conformance
When integrated with medical device manufacturing software and medical device erp systems, organizations shift from reactive correction to proactive risk control protecting revenue and sustaining operational continuity.
Driving Continuous Improvement Across the Organization
Compliance software is not static. It feeds continuous improvement loops by connecting:
- Quality events from production
- Risk data from design and field performance
- Audit findings and CAPA outcomes
This closed-loop system ensures that insights translate into measurable improvements across design, manufacturing, and supplier management. Medical device compliance software plays a central role in maintaining regulatory alignment, supporting audit readiness, and reducing financial exposure. Integrated with medical device erp software, ERP for medical device manufacturers, and medical device manufacturing software, it enables organizations to operate with confidence in a tightly regulated environment where compliance is enforced through systems, not left to interpretation.

Medical Equipment Management Software: Keeping Assets Safe, Calibrated, and Accounted For
Hospitals operate under constant pressure to maintain clinical continuity. When equipment is not tracked, maintained, and calibrated within controlled systems, the impact is immediate procedure delays, patient safety incidents, and unplanned capital expenditure. Medical equipment management software forms the operational foundation that prevents these failures, ensuring that assets remain available, compliant, and financially accountable.
Without this layer, even the most sophisticated medical device manufacturing software and medical device erp software lose downstream visibility once devices enter hospital environments. Asset control must continue beyond procurement.
The Hidden Cost of Unmanaged Equipment
Untracked or poorly maintained devices introduce risk across multiple dimensions:
- Equipment downtime disrupts clinical schedules and revenue cycles
- Calibration gaps compromise diagnostic accuracy and treatment outcomes
- Emergency replacements inflate capital budgets
- Lack of visibility leads to over-purchasing or under utilization
Medical equipment management software addresses these gaps by maintaining a live registry of assets, service history, and performance metrics ensuring that decisions are based on data, not assumptions.
Core Capabilities That Define Equipment Management
1. Asset Registry and Identification
Each device is recorded with serial numbers, location data, ownership details, and service history. This registry becomes the single reference point for tracking equipment across departments.
2. Maintenance Scheduling and Work Orders
Preventive maintenance schedules are enforced within the system, ensuring that servicing occurs within defined intervals. Work orders are generated, tracked, and documented to maintain accountability.
3. Calibration Tracking
Calibration records are maintained with expiry alerts and validation logs. This ensures compliance with clinical standards and prevents the use of inaccurate equipment in patient care.
4. Lifecycle Management
From procurement to decommissioning, medical equipment management software tracks asset performance, repair frequency, and cost trends—supporting informed replacement decisions.
Why Hospital Equipment Management Software Is Different
Generic asset management tools lack the controls required in clinical environments. Hospitals require:
- Calibration traceability linked to clinical usage
- Maintenance records aligned with regulatory audits
- Integration with procurement and supplier data
- Risk-based prioritization for critical equipment
Medical equipment management software integrates with medical equipment inventory management software and upstream systems such as medical device ERP systems, ensuring that asset data remains consistent from manufacturer to hospital floor.
Regulatory Alignment Is Mandatory
Healthcare facilities must comply with standards enforced by organizations such as The Joint Commission, DNV, and Centers for Medicare & Medicaid Services.
These frameworks require:
- Documented maintenance schedules
- Calibration records for critical devices
- Evidence of inspection and performance checks
- Risk-based equipment management plans
Medical equipment management software ensures that this documentation is maintained, accessible, and audit-ready at any point.
Integration with Clinical Systems
Asset performance must align with clinical demand. Integration with hospital systems enables:
- Utilization tracking across departments
- Identification of underused or overburdened equipment
- Data-driven decisions on asset allocation and procurement
This integration connects hospital operations with erp for medical device manufacturers, creating a feedback loop that informs production, supply planning, and service models.
Mobile-Enabled Workflows for Field Technicians
Technicians operate across wards, labs, and operating rooms. Mobile-enabled medical equipment management software allows them to:
- Access asset records on-site
- Update maintenance logs in real time
- Capture inspection data with timestamps and user authentication
This reduces administrative overhead and ensures that service data is recorded at the point of activity.
Benchmarking Performance Across the Asset Base
Hospitals require measurable indicators to maintain operational control. Medical equipment management software tracks:
- Equipment uptime rates
- Mean Time Between Failures (MTBF)
- Maintenance cost per asset
- Service response times
These metrics support performance benchmarking, budget planning, and continuous improvement across biomedical engineering teams. Medical equipment management software anchors hospital operations in control, accountability, and compliance. Integrated with medical equipment inventory management software, medical device erp software, and upstream medical device manufacturing software, it ensures that devices remain safe, available, and aligned with clinical and regulatory expectations throughout their lifecycle.
CMMS for Hospitals: The Maintenance Backbone Every Biomed Team Needs
A Computerised Maintenance Management System (CMMS) is the execution layer that keeps hospital equipment operational, compliant, and cost-controlled. Medical equipment management software defines the asset landscape; CMMS drives the day-to-day maintenance activity that sustains uptime. For biomedical engineering and facilities teams, this is where service work is planned, tracked, and validated. Unlike general asset tools that focus on inventory visibility, CMMS governs maintenance workflows, technician activity, and service documentation with audit-ready discipline. Integrated with medical equipment inventory management software and upstream medical device erp software, CMMS ensures that maintenance data feeds procurement, budgeting, and compliance records across the organization.
What CMMS Does and Why It Differs from Asset Management
Asset management platforms answer “what do we own and where is it located.”
CMMS answers “what work must be performed, when, by whom, and at what cost.”
This distinction matters in clinical environments where maintenance execution determines equipment availability and regulatory compliance. CMMS systems enforce service schedules, track work orders, and maintain detailed service histories capabilities that general tools cannot support at the required level.
Preventive Maintenance Scheduling That Reduces Risk
CMMS platforms enforce maintenance schedules using multiple trigger models:
- Interval-based scheduling: service events triggered by time cycles (monthly, quarterly, annual)
- Meter-based scheduling: maintenance driven by usage metrics such as operating hours or cycles
- Condition-based triggers: service initiated based on performance indicators or inspection results
These mechanisms ensure that critical devices remain calibrated and operational, aligning with hospital safety standards and manufacturer recommendations supported by medical device manufacturing software.
Work Order Management That Drives Accountability
Maintenance execution is controlled through structured work orders within CMMS:
- Creation of service requests from scheduled plans or incident reports
- Assignment to technicians with defined priorities
- Tracking of spare parts consumption and inventory linkage
- Recording of labour time and service notes
- Controlled closure with verification and documentation
This workflow ensures that no maintenance activity is missed, undocumented, or delayed supporting compliance and operational continuity.
Managing Biomedical and Facilities Maintenance in One Platform
Hospitals maintain two distinct asset categories:
- Biomedical equipment: diagnostic and therapeutic devices requiring calibration and regulatory oversight
- Facilities infrastructure: HVAC systems, electrical networks, and building assets
A unified CMMS enables both domains to be managed within one environment, ensuring consistent workflows, shared reporting, and consolidated cost tracking. For larger organizations, separation may exist; however, integration between systems remains essential to maintain visibility across the asset base.
Compliance Alignment with Equipment Management Standards
Accreditation bodies such as The Joint Commission enforce strict equipment management requirements, including standards like EC.02.04.01.
CMMS supports compliance by:
- Maintaining documented maintenance schedules
- Recording completed service activities with timestamps
- Providing audit trails for inspections and corrective actions
- Demonstrating adherence to risk-based maintenance strategies
This documentation is essential during audits and inspections, ensuring that hospitals can validate equipment safety and readiness.
Parts and Labour Cost Tracking — Building the Financial Case
CMMS platforms track:
- Spare parts usage linked to each work order
- Labour hours by technician and task
- Service frequency and repair patterns per asset
This data enables hospitals to evaluate:
- Whether in-house maintenance delivers cost advantages
- When outsourcing service contracts is justified
- Which assets generate disproportionate maintenance costs
When connected with the medical device ERP system, these insights influence procurement strategies, budgeting, and vendor negotiations.
Integration Across the Hospital Technology Stack
CMMS does not operate in isolation. Integration ensures continuity across systems:
- CMMS ↔ Medical Equipment Management Software: asset data, maintenance schedules, and service history
- CMMS ↔ Medical Equipment Inventory Management Software: spare parts availability and stock control
- CMMS ↔ Procurement Systems: automated reordering of critical components
- CMMS ↔ Medical Device ERP Software: financial tracking, supplier data, and compliance records
This connected environment ensures that maintenance activity contributes to organization-wide visibility and control. CMMS is the operational engine behind hospital maintenance. Integrated with medical equipment management software, medical equipment inventory management software, and upstream medical device erp software, it ensures that biomedical teams maintain equipment reliability, meet regulatory expectations, and control service costs with confidence.
Medical Equipment Inventory Management: From Spreadsheets to Real-Time Visibility
Hospitals manage thousands of assets, consumables, and implantable devices across departments. When inventory is tracked through disconnected tools, visibility breaks down driving excess stock, stockouts, and financial leakage. Industry data shows that 30–40% of hospital supply chain costs can be traced to poor inventory control. Medical equipment inventory management software replaces fragmented tracking with controlled, system-driven visibility. Integrated with medical equipment management software and upstream medical device erp software, it establishes accountability across procurement, storage, usage, and replenishment.
The Inventory Accuracy Challenge
Inventory gaps do not remain isolated, they impact clinical delivery, procurement cycles, and financial reporting. Common issues include:
- Missing or unaccounted equipment across departments
- Overstocking driven by lack of demand visibility
- Critical stockouts affecting procedures
- Inconsistent reconciliation between physical stock and purchase records
Medical equipment inventory management software addresses these issues by maintaining continuous visibility across locations, ensuring that inventory data reflects actual usage and availability.
Tracking Serialized Medical Equipment Across the Hospital
Hospitals require granular tracking of high-value assets. Medical equipment inventory management software enables:
- Serial-level tracking for devices across wards, labs, and operating rooms
- Location mapping with movement history
- Ownership and department-level allocation
- Integration with maintenance records from medical equipment management software
This ensures that equipment remains traceable, auditable, and aligned with operational needs.
Managing Medical Supplies and Consumables at Scale
Beyond equipment, hospitals must control consumables such as surgical kits, implants, and disposables. Inventory systems support:
- Par-level automation to maintain optimal stock levels
- Usage tracking tied to procedures and departments
- Kit assembly and consumption tracking
- Replenishment triggers based on demand patterns
This level of control ensures that high-cost items are available when required without tying up excess capital.
RFID and Barcode Scanning: Choosing the Right Tracking Model
Tracking technologies define how inventory data is captured:
- Barcode scanning: cost-effective, suitable for controlled environments where staff scan items during movement or usage
- Passive RFID: enables bulk scanning without line-of-sight, improving efficiency in storage areas
- Active RFID: provides continuous location tracking for critical assets, supporting high-value equipment management
Hospitals adopt active RFID when the cost of asset loss, misplacement, or downtime exceeds the investment in continuous tracking.
Expiry Management with FEFO Logic
Expired supplies create both financial loss and clinical risk. Medical equipment inventory management software enforces First-Expiry, First-Out (FEFO) logic:
- Prioritizing usage of items nearing expiry
- Generating alerts for upcoming expirations
- Preventing issuance of expired stock
This is essential for implantables, pharmaceuticals, and critical consumables where safety and compliance are non-negotiable.
Vendor-Managed Inventory (VMI): Extending Control to Suppliers
In high-volume or high-value categories, hospitals adopt Vendor Managed Inventory models. Suppliers monitor stock levels and manage replenishment based on agreed thresholds.
Medical equipment inventory management software enables VMI by:
- Sharing inventory data with approved vendors
- Tracking supplier performance and fill rates
- Maintaining audit trails for replenishment decisions
This reduces administrative burden while maintaining accountability and control.
Closing the Loop with Procurement and ERP Systems
Inventory visibility must align with financial and procurement systems. Integration ensures:
- Purchase orders generated based on actual demand
- Goods receipt matched with inventory records
- Automated reconciliation between physical stock and financial data
When connected with the medical device ERP system and ERP for medical device manufacturers, hospitals achieve closed-loop control from procurement to consumption ensuring that inventory decisions are financially and operationally aligned. Medical equipment inventory management software transforms hospital supply chains from reactive tracking to controlled visibility. Integrated with medical equipment management software, medical device manufacturing software, and medical device ERP software, it ensures that equipment and supplies remain available, traceable, and aligned with clinical and financial priorities.

Hospital Pharmacy Inventory Management Software: Precision Where It Matters Most
Pharmacy operations carry the highest level of regulatory scrutiny and patient risk within the hospital supply chain. Controlled substances, temperature-sensitive drugs, and high-cost therapies demand strict oversight. Medical equipment inventory management software alone cannot address these requirements. Hospitals require dedicated pharmacy systems that enforce traceability, compliance, and controlled dispensing at a granular level. For pharmacy directors and supply chain leaders, this is where inventory integrity translates into patient safety, regulatory compliance, and financial control.
Why Pharmacy Inventory Requires a Dedicated System
Pharmacy environments introduce risks that general inventory platforms cannot manage:
- Controlled substances requiring strict documentation and reconciliation under Drug Enforcement Administration oversight
- Cold chain requirements for temperature-sensitive medications
- Look-alike and sound-alike (LASA) drugs that increase dispensing error risk
- High-value inventory with tight expiry windows
Dedicated pharmacy inventory systems enforce safeguards across these dimensions, ensuring that drug handling aligns with clinical and regulatory expectations.
Core Capabilities That Define Pharmacy Inventory Systems
1. Formulary Management
Hospitals maintain controlled drug lists aligned with clinical protocols and cost strategies. Pharmacy systems ensure that procurement and dispensing adhere to approved formularies, reducing variation and cost leakage.
2. Automated Dispensing Cabinet (ADC) Integration
Integration with ADCs ensures controlled access to medications at the point of care. Inventory updates reflect actual dispensing activity, maintaining alignment between central pharmacy stock and ward-level usage.
3. 340B Program Tracking
For eligible hospitals, tracking under 340B Drug Pricing Program ensures compliance with pricing rules and audit requirements. Systems maintain eligibility records, purchase tracking, and reporting accuracy.
Controlled Substance Reconciliation and Diversion Detection
Controlled drug management requires strict accountability. Pharmacy inventory systems:
- Track dispensing and returns at user level
- Reconcile physical stock against system records
- Flag discrepancies for investigation
- Support diversion detection through usage pattern analysis
These controls ensure compliance with regulatory expectations and protect against misuse or loss.
Expiry and Recall Management at Batch Level
Drug safety depends on full traceability. Systems maintain:
- Batch and lot-level tracking from manufacturer to patient
- Expiry monitoring with automated alerts
- Recall management workflows to identify and isolate affected stock
When integrated with medical device manufacturing software and medical device erp software, hospitals gain visibility into upstream production and recall notifications, enabling faster response.
Analytics That Drive Clinical and Financial Outcomes
Pharmacy inventory systems provide actionable insights:
- Drug utilization review to monitor prescribing patterns
- Formulary adherence tracking to enforce clinical guidelines
- Wastage analysis to reduce expired or unused stock
These insights allow pharmacy leaders to balance patient care quality with cost control.
Integration Across Hospital Systems
Pharmacy operations must align with clinical and financial systems. Integration ensures:
- HIS and EHR connectivity for prescription validation and patient-level tracking
- Procurement platform linkage for demand-driven purchasing
- Medical equipment management software alignment for storage and handling equipment
- Medical device erp system integration for financial reconciliation and supplier coordination
This connected environment ensures that pharmacy data flows across the hospital ecosystem without gaps.
Measurable Cost Impact
Drug spend represents a significant portion of hospital budgets. Implementation of pharmacy inventory systems delivers measurable outcomes:
- Reduction in excess inventory through demand alignment
- Lower wastage driven by expiry control
- Improved pricing compliance through program tracking
- Strengthened audit readiness reducing financial penalties
Hospitals report drug spend reductions in the range of 8–15% when pharmacy inventory is managed through controlled systems rather than fragmented tools. Hospital pharmacy inventory management software establishes control where risk is highest. Integrated with medical equipment inventory management software, medical equipment management software, and upstream medical device erp software, it ensures that medications remain safe, traceable, and aligned with regulatory and financial expectations across the hospital environment.
Medical Laboratory Management System: Where Data Quality Begins
Laboratories operate at the junction of clinical care and supply chain control. Reagents, instruments, specimens, and diagnostic results move through tightly governed workflows where accuracy is non-negotiable. A medical laboratory management system (LIMS) ensures that data integrity, traceability, and compliance are enforced from sample intake through result delivery. For hospitals and diagnostic centers, LIMS connects laboratory operations with medical equipment management software, medical equipment inventory management software, and upstream medical device ERP software creating a controlled environment where clinical decisions rely on validated data.
What a Medical Laboratory Management System Manages
LIMS extends far beyond test result reporting. It governs the complete laboratory workflow:
- Sample registration and accessioning
- Test order management and workflow routing
- Instrument data capture and validation
- Quality control tracking and exception handling
- Result verification, approval, and release
This ensures that each diagnostic output is backed by traceable processes and validated inputs.
Inventory Control Within the Laboratory
Laboratory inventory requires tight oversight due to sensitivity and cost. LIMS manages:
- Reagent inventory: tracking stock levels, usage, and replenishment cycles
- Calibrator and control materials: ensuring availability for test validation
- Quality control (QC) lot management: linking QC data to test results for audit traceability
Integrated with medical equipment inventory management software, this ensures that laboratory materials remain aligned with testing demand and compliance requirements.
Instrument Integration Eliminates Manual Errors
Modern laboratories depend on analyzers and diagnostic instruments. LIMS integrates with these systems to:
- Capture test results directly from instruments
- Eliminate transcription errors associated with manual data entry
- Maintain data consistency across test workflows
This integration strengthens data accuracy and supports high-throughput environments where manual processes introduce risk.
Compliance and Audit Readiness
Laboratories must comply with standards enforced by CLIA, College of American Pathologists, and ISO 15189.
LIMS supports compliance by:
- Maintaining validated test methods and documentation
- Recording quality control results and deviations
- Generating audit trails for all laboratory activities
- Ensuring that only authorized personnel perform and approve tests
This ensures that laboratories remain inspection-ready at any point.
Chain of Custody for Specimen Tracking
Specimen integrity is critical. LIMS enforces chain-of-custody tracking:
- From sample collection and labeling
- Through transport and processing
- To analysis and final result release
Each step is time stamped and user validated, ensuring traceability and accountability across the testing lifecycle.
Integration with Hospital Systems
LIMS operates within a connected hospital ecosystem. Integration enables:
- Bi-directional communication with hospital information systems for test orders and results
- Alignment with electronic health records for patient-level data
- Connectivity with medical device erp system for procurement and supplier data
- Coordination with medical equipment management software for instrument maintenance and calibration
This ensures that laboratory data flows across clinical and operational systems without gaps.
Turnaround Time Analytics and Process Optimization
Timely diagnostics impact patient outcomes. LIMS provides analytics to:
- Measure turnaround time across test categories
- Identify bottlenecks in sample processing or instrument availability
- Optimize workflow sequencing and resource allocation
These insights enable laboratories to improve efficiency, maintain service levels, and support clinical decision-making with timely results. A medical laboratory management system establishes control at the point where diagnostic data originates. Integrated with medical equipment inventory management software, medical equipment management software, and medical device manufacturing software, it ensures that laboratory operations remain accurate, traceable, and aligned with clinical and regulatory expectations.
Building an Integrated Healthcare Software Stack: From Siloed Tools to Connected Operations
Disconnected systems create blind spots. In regulated healthcare environments, those gaps translate into compliance exposure, operational delays, and financial leakage. A connected stack anchored by medical device manufacturing software, medical device erp software, and medical equipment management software establishes continuity across production, supply chain, and clinical operations. This section outlines how to connect these systems, sequence implementation, and evaluate vendors with a compliance-first lens.
The Integration Imperative
Standalone tools solve isolated problems but introduce fragmentation:
- Duplicate data across systems
- Inconsistent audit trails
- Delayed decision-making due to lack of visibility
An integrated environment connects medical device erp system, medical device manufacturing software, medical equipment inventory management software, and compliance platforms into a validated data flow. This ensures that product data, quality records, and operational metrics remain aligned across the lifecycle—from manufacturing to patient use.
Recommended Implementation Sequence for Hospitals
A phased rollout reduces disruption and strengthens adoption:
1. CMMS (Maintenance Execution Layer)
Establish maintenance control and service documentation across biomedical and facilities teams.
2. Medical Equipment Management Software
Create a governed asset registry with maintenance, calibration, and lifecycle tracking.
3. Medical Equipment Inventory Management Software
Introduce inventory visibility for equipment and consumables, enabling demand-driven replenishment.
4. Pharmacy Inventory Systems
Extend control to high-risk drug inventory with compliance and dispensing safeguards.
5. Laboratory Management System (LIMS)
Integrate diagnostics, reagent tracking, and result workflows into the broader ecosystem.
This sequence ensures that foundational controls are established before expanding into high-complexity domains.
Recommended Stack for Device Manufacturers
For manufacturers, the priority is compliance-driven production and traceability:
1. Medical Device ERP Software
Establish the system of record for product data, supplier management, and financial control.
2. Medical Device Manufacturing Software (MES)
Control production execution, batch traceability, and quality enforcement.
3. Medical Device Compliance Software
Manage documentation, CAPA workflows, audit readiness, and regulatory alignment.
4. Supplier Portal Integration
Extend visibility to vendors for quality data exchange, inspection results, and corrective actions.
This stack ensures that erp for medical device manufacturers connects upstream supplier data with downstream production and compliance processes.
API Standards That Enable Interoperability
Interoperability depends on standardized data exchange:
- HL7 FHIR for clinical systems, enabling structured exchange between hospital platforms, EHRs, and laboratory systems
- REST APIs for system-to-system communication across ERP, inventory, and maintenance platforms
- EDI protocols for procurement, supplier transactions, and logistics integration
These standards ensure that systems communicate without data loss or inconsistency.
Cloud vs. On-Premise: The Validation and Security Equation
Deployment decisions now revolve around validation, scalability, and data governance:
- Cloud environments provide faster deployment, centralized updates, and scalable infrastructure. Validation approaches align with evolving regulatory expectations for system assurance.
- On-premise environments offer infrastructure control and internal governance, suitable for organizations with established validation frameworks.
Across both models, medical device ERP systems and medical device manufacturing software must maintain validated states, controlled updates, and documented change management aligned with regulatory requirements.
Change Management: The Hidden Risk Factor
Technology does not fail, adoption does. Organizations underestimate:
- Training requirements across departments
- Resistance from operational teams
- Misalignment between IT and clinical stakeholders
Successful implementation requires:
- Executive sponsorship
- Defined ownership across functions
- Continuous training and performance tracking
Without these elements, even well-designed systems fail to deliver expected outcomes.
Frequently Asked Questions
1. What is the role of medical device ERP software in a healthcare stack?
Medical device erp software serves as the system of record that governs product, supplier, financial, and compliance data across the organization. It connects upstream manufacturing inputs with downstream hospital usage, ensuring that data remains consistent, traceable, and audit-ready.
Within ERP for medical device manufacturers, key functions include:
- Product structure and BOM control linked to design and production
- Supplier qualification, incoming inspection, and corrective action tracking
- Financial oversight including lot-level costing, warranty provisions, and recall exposure
- Regulatory documentation aligned with standards such as FDA 21 CFR Part 820 and ISO 13485
When integrated with medical device manufacturing software and compliance platforms, the medical device ERP system ensures that data flows across departments without duplication or gaps, supporting inspection readiness and operational control.
2. How does medical device manufacturing software integrate with hospital systems?
Medical device manufacturing software connects to hospital environments through the medical device erp system and interoperability standards such as HL7 FHIR. This integration ensures continuity from production to clinical use.
Key integration points include:
- Batch and serial traceability flowing into medical equipment inventory management software
- Device identification and labeling aligned with UDI requirements
- Recall and field safety data shared with hospital systems for rapid response
- Maintenance and performance feedback from medical equipment management software informing manufacturing improvements
This closed-loop connection enables manufacturers to track device performance in real-world settings and allows hospitals to maintain full visibility into device origin, configuration, and compliance status.
3. Why is medical equipment inventory management software critical for hospitals?
Medical equipment inventory management software provides controlled visibility across high-value assets, consumables, and implantable devices. Without it, hospitals face stock inconsistencies, financial leakage, and compliance exposure.
Core value drivers include:
- Serial-level tracking of equipment across departments and locations
- Demand-driven replenishment for consumables and surgical kits
- Expiry control using FEFO logic to reduce wastage
- Integration with procurement and medical device erp software for accurate financial reconciliation
This system works alongside medical equipment management software to ensure that assets are not only visible but also maintained, calibrated, and available for clinical use. The result is improved operational continuity, reduced emergency purchases, and stronger audit readiness.
4. What is the difference between CMMS and medical equipment management software?
CMMS and medical equipment management software serve related but distinct roles within hospital operations:
- CMMS (Computerised Maintenance Management System):
Focuses on maintenance execution work orders, technician assignments, spare parts usage, and service documentation. It ensures that preventive and corrective maintenance activities are completed and recorded. - Medical Equipment Management Software:
Governs the broader asset lifecycle, including asset registry, calibration tracking, compliance documentation, and performance benchmarking. It provides a strategic view of asset utilization and risk.
In practice, CMMS operates as the execution engine within the framework defined by medical equipment management software. When integrated with medical equipment inventory management software and medical device ERP systems, these tools create a connected environment where asset performance, maintenance activity, and financial data remain aligned.
5. How should organizations prioritize implementation?
Implementation sequencing determines adoption success and long-term ROI. A structured rollout reduces disruption and ensures that foundational controls are in place before expanding into complex domains.
For Hospitals:
- Start with CMMS to establish maintenance discipline and service tracking
- Deploy medical equipment management software to build a governed asset registry
- Introduce medical equipment inventory management software for inventory visibility and replenishment control
- Extend into pharmacy inventory systems for high-risk drug management
- Implement laboratory systems to connect diagnostics, reagents, and result workflows
For Manufacturers:
- Begin with medical device erp software to establish the system of record
- Add medical device manufacturing software for production control and traceability
- Implement compliance platforms to manage documentation, CAPA, and audit readiness
- Integrate supplier portals for quality collaboration and data exchange
This phased approach ensures that ERP for medical device manufacturers, medical device manufacturing software, and hospital-side systems evolve into a connected, compliant ecosystem supporting traceability, operational control, and sustained performance across the healthcare value chain.
6. How easy is it to train people on Odoo ERP
Odoo ERP for SMEs is designed to help users learn the application quickly.
- Comprehensive Documentation: Odoo provides extensive online resources, tutorials, and manuals to guide users through functions and features.
- Customizable Dashboards: Personalized dashboards help users focus on relevant tasks, reducing the complexity of learning multiple features.
Interactive Community - Support: A large Odoo community offers forums, webinars, and peer support, helping users resolve issues and learn quickly.
- Training Programs and Certifications: Odoo offers structured training and certification courses to ensure in-depth knowledge transfer.
- Training from the Implementation Partner: Once deployed, ensure your implementation partner trains your teams in areas where Odoo is customized.
- Hands-on Learning: Odoo’s live demos and test environments provide users with practical experience and allow them to learn by doing.
Schedule a conversation with us now!
written by

Abinay Subramaniam P
ERP Project Manager
Abinay Subramaniam P is a Project Manager at Navabrind IT Solutions with a proven track record across several verticals, including Manufacturing, Publishing, and IT. He excels in high‑pressure, fast‑paced software development environments, consistently delivering solutions that exceed customer expectations. Abinay brings strong expertise in negotiation, client engagement, and team management, ensuring smooth project execution and stakeholder alignment. He is trained in PMP and holds the Certified Scrum Master (CSM) credential, combining structured project management practices with agile methodologies.
Related Articles
-
Post
Why Indian Manufacturers Are Losing Lakhs Every Year Without ERP Software
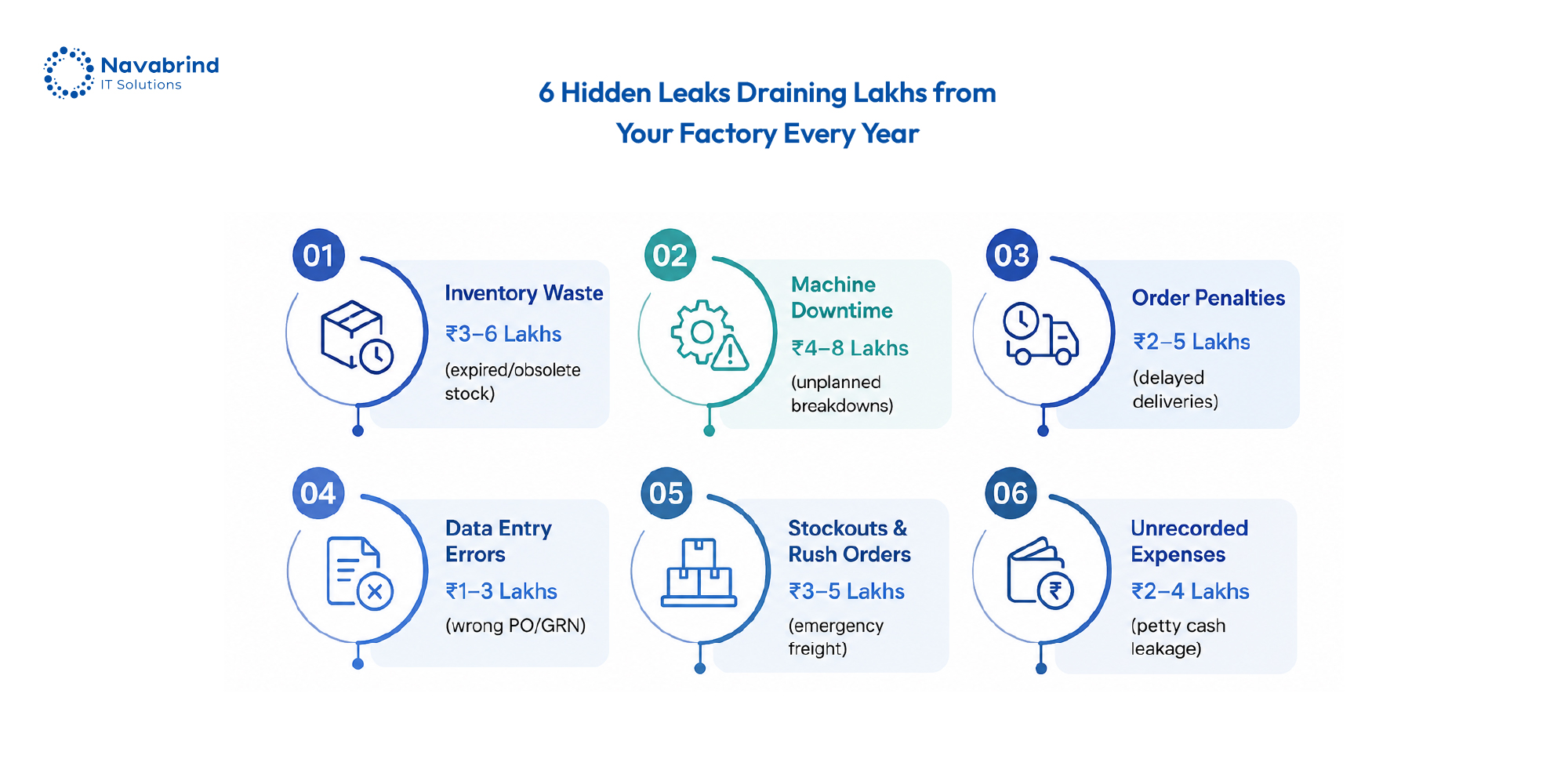
Why Indian Manufacturers Are Losing Lakhs Every Year Without ERP Software June 1, 2026 Posted by: Jaishree Jayabal Singh Categories: Blog, Odoo ERP for Manufacturing No Comments Indian manufacturers are under pressure from rising material costs, tighter delivery timelines, GST compliance demands, machine downtime, and unpredictable supply chains. Yet many factories still depend on disconnected -
Post
How Odoo ERP Integration Improves Business for US Companies
How Odoo ERP Integration Improves Business for US Companies May 29, 2026 Posted by: Jaishree Jayabal Singh Categories: Blog, Business Automation, ERP Integration, Odoo ERP No Comments US companies face a growing technology strain: disconnected applications that perform individual tasks yet fracture business visibility. Teams juggle CRM, finance, eCommerce, inventory, logistics, and support platforms, each -
Post
5 Reasons Indian Retail Businesses Are Struggling With Inventory in 2026 (And How to Fix It)
5 Reasons Indian Retail Businesses Are Struggling With Inventory in 2026 (And How to Fix It) May 28, 2026 Posted by: Dania Sibionna Philemon Categories: Blog, Inventory Management, Odoo ERP, Retail ERP No Comments The hidden cost of poor inventory management in Indian retail dead stock, stockouts, shrinkage, and lost sales Inventory errors drain profit
written by

Abinay Subramaniam P
ERP Project Manager
Abinay Subramaniam P is a Project Manager at Navabrind IT Solutions with a proven track record across several verticals, including Manufacturing, Publishing, and IT. He excels in high‑pressure, fast‑paced software development environments, consistently delivering solutions that exceed customer expectations. Abinay brings strong expertise in negotiation, client engagement, and team management, ensuring smooth project execution and stakeholder alignment. He is trained in PMP and holds the Certified Scrum Master (CSM) credential, combining structured project management practices with agile methodologies.
How can we help you?
Get in touch with a solutions consultant that can share best practices and help solve specific challenges.